Enabling Early Detection for Healthier Brain
Revolutionizing neurology diagnostics with our digital NeuroMarker Platform designed to rapidly detect early cognitive decline and empower healthcare providers to intervene sooner—when treatment is most effective.
Learn more about our platform
Moving Beyond Traditional Cognitive Assessments
Traditional pen-and-paper assessments for Mild Cognitive Impairment (MCI) lack sensitivity, require specialists, and take hours, often missing critical treatment windows. First-generation digital assessments merely digitize these outdated methods. Altoida’s NeuroMarker Platform transforms the process with neuroscience-driven tasks that simulate daily activities, capturing motion, speech, and touch data for deeper insights. The self-administered, in-clinic Altoida Digital Cognitive Assessment provides rapid, objective results in just 10 minutes.

Streamlining Neurological
Diagnostics for Faster Interventions
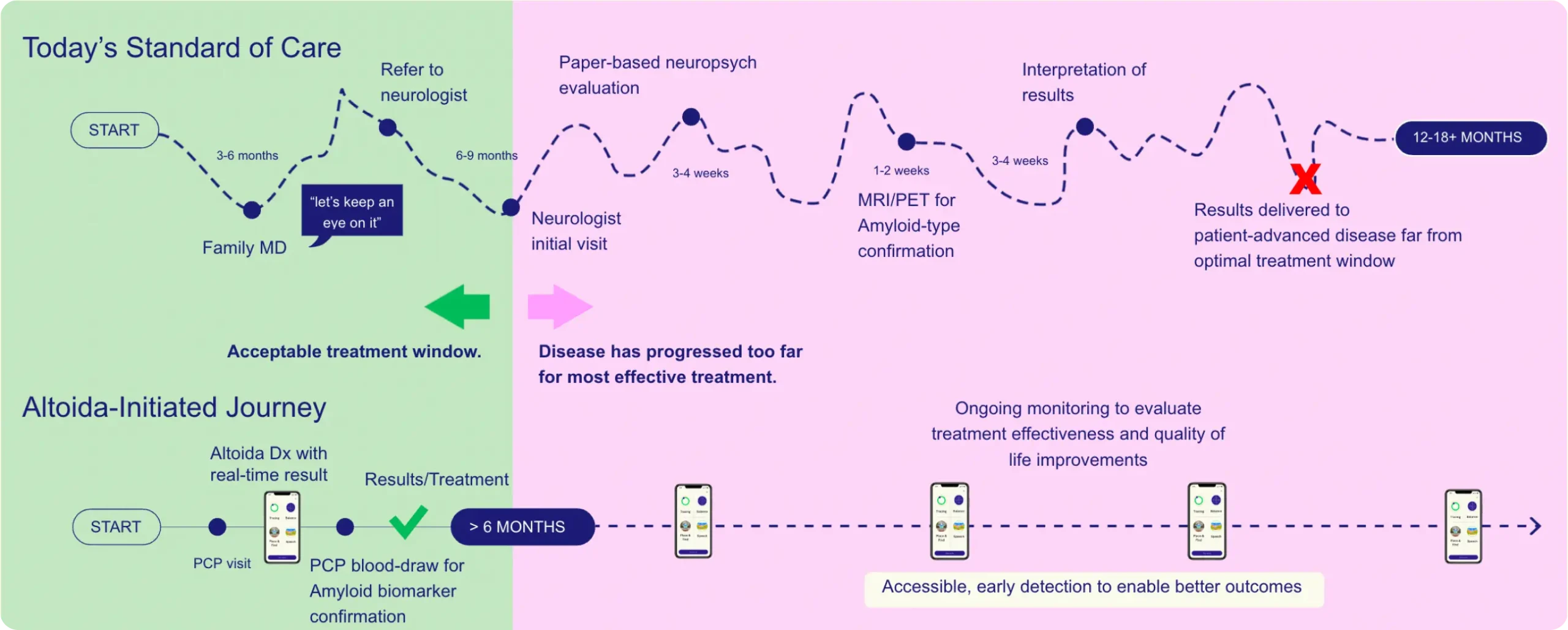
Current Diagnostics Miss Early Detection Leading to Long Wait Times for Treatment Decisions
Pen and Paper Assessment
2–4 hours of testing with a trained neuropsychologist.
Lacks the sensitivity to detect early MCI, delaying care.
Months to interpret & develop a treatment plan.
Altoida NeuroMarker Platform
10-minute self-administered assessment in a single clinic visit.
Uses neuroscience-driven tasks & sensor-based data for highly sensitive early detection.
Real-time, objective results, enabling clinicians to make decisions quickly.
Outperforming Standard of Care by Addressing Critical Gaps in Early Detection
Standard of Care

Assessment Type
Up to 4 hours In-clinic
As little as 10 minutes In-clinic
Administration
Trained healthcare professional
Self-administered
Patient-centric
Early and reliable identification of MCI
Robust data assesses cognitive status
Designed for primary care
Enhance Care with Advanced Cognitive Diagnostics
The Altoida NeuroMarker Platform is designed to:

Real-Time Cognition Monitoring
Enable continuous, real-time monitoring
of cognitive function, allowing for early
detection of changes.
of cognitive function, allowing for early
detection of changes.

Personalize Treatment Paths
Guide healthcare providers in personalizing treatment plans to address specific areas of cognitive decline or impairment.

Monitor Real-Time Progress
Track changes over time, enabling clinicians to evaluate the progression of cognitive disorders and make informed decisions about medication adjustments and other care strategies.

Enhance Patient Engagement
Understanding of their condition, encouraging adherence to treatment plans as patients observe their progress.
Altoida in Action
Watch how Altoida’s Digital Cognitive Assessment puts patients at the center of their cognitive health journey. In just 10 minutes, patients can take a simple, non-invasive test using a smart tablet designed to enable early detection of MCI.
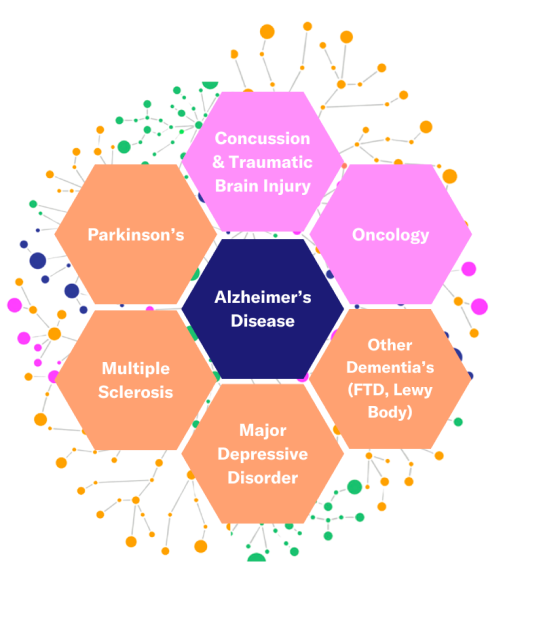
Mild Cognitive Impairment is Just the Start
Building on two decades of scientific research, Altoida’s NeuroMarker Platform combines advanced neuroscience, machine learning, augmented reality, and sensor data to deliver deep, evidence-based insights into brain health. Designed to move beyond traditional assessments, it sets a new standard for neurological diagnostics, starting with MCI and Alzheimer’s, while expanding into other central nervous system diseases. Our innovative approach ensures every assessment is transparent, measurable, and grounded in cutting-edge science, paving the way for advancements in cognitive health.
Get in Touch with Us
Whether you have questions about our platform or want to explore partnership opportunities, we’re here to help. Our team is ready to provide you with the answers and support you need.
Our platform is currently intended for investigational use only. The platform has not received FDA clearance or approval and is not available for commercial sale.
